Asbury Communities has promoted and implemented the best evidence-based practices in mitigating the COVID-19 risk since the beginning of the pandemic. We have consistently aligned our practice with the expertise and guidance of the Centers for Disease Control & Prevention (CDC). As always, we are grateful for your continued vigilance and your support in keeping each other safe during this pandemic.
See Frequently Asked Questions below our Associate Vaccine Photos.
See Vaccine Clinic Dates below.
Vaccine processes, eligibility, and supplies are set by each state and they vary. Asbury is working aggressively to secure vaccines for all those who live and work at our communities and wish to be vaccinated. To date, Asbury has vaccinated 95% of the residents living at our communities and 70% of the associates working at our communities.




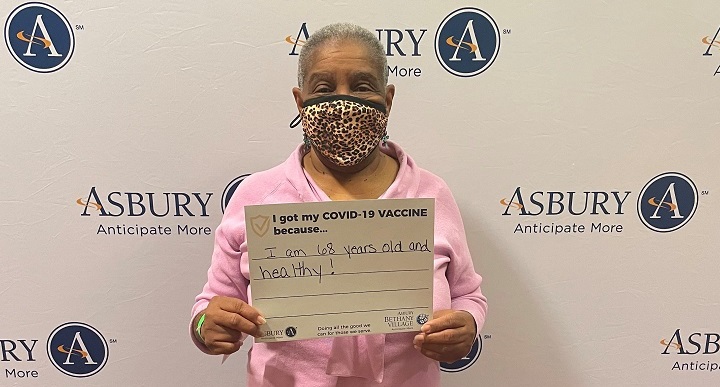









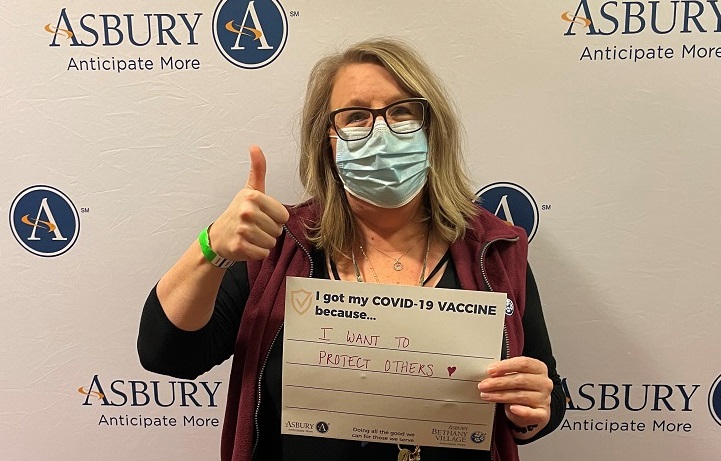


















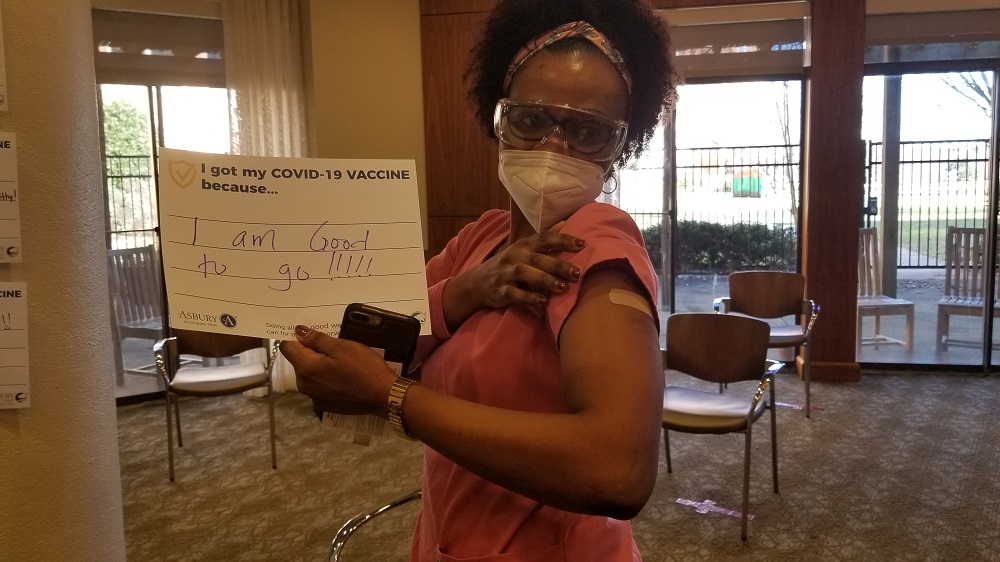
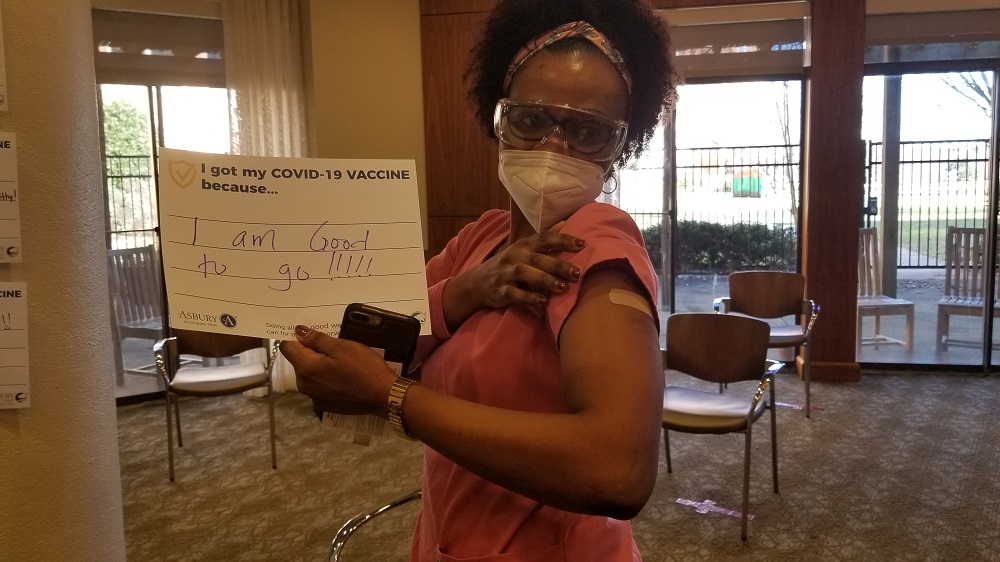


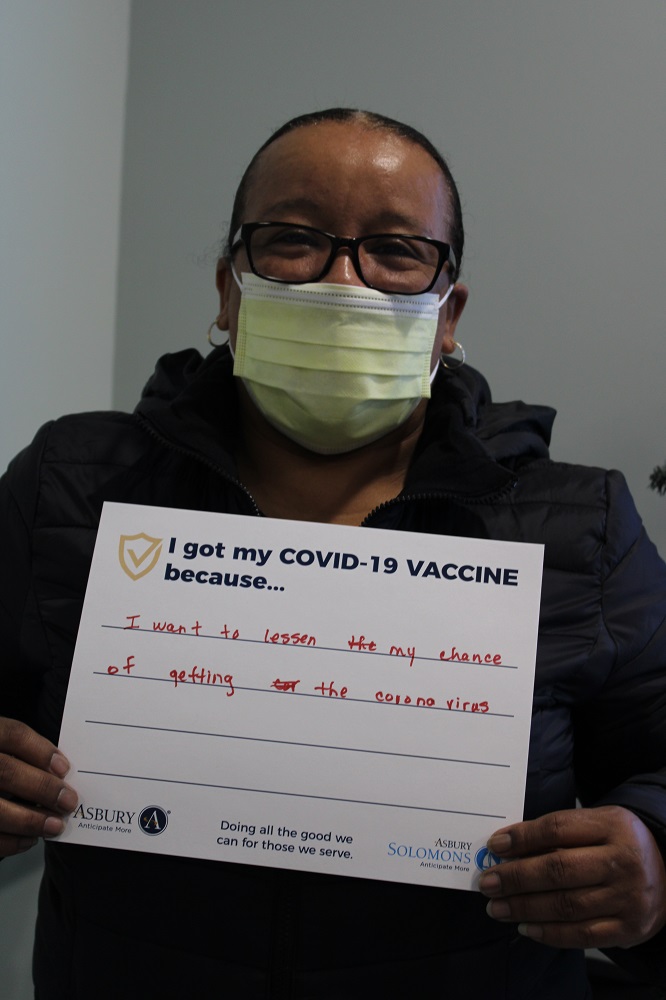
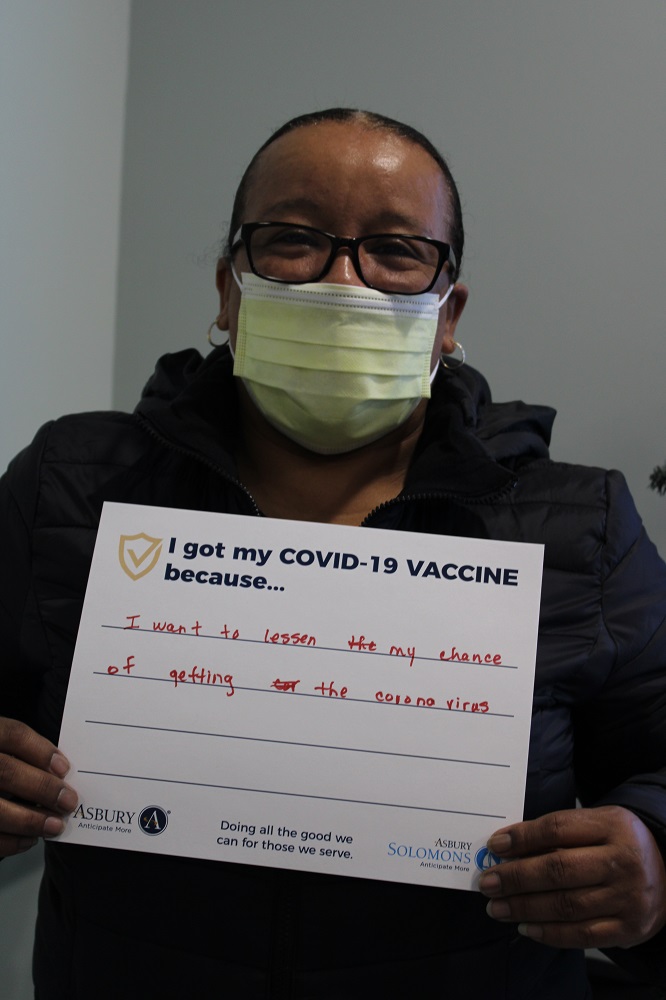






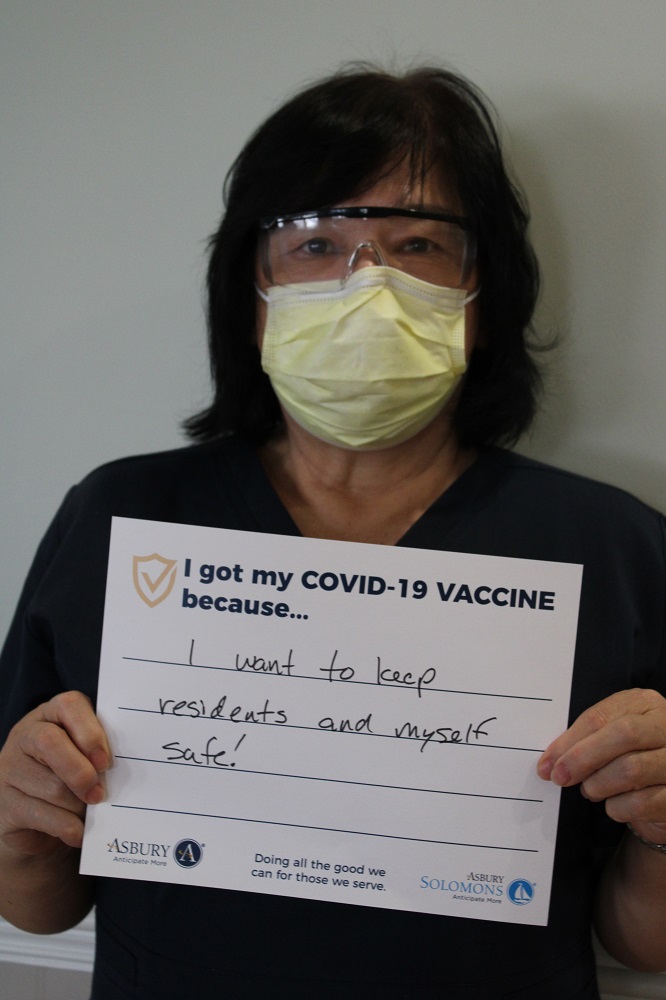







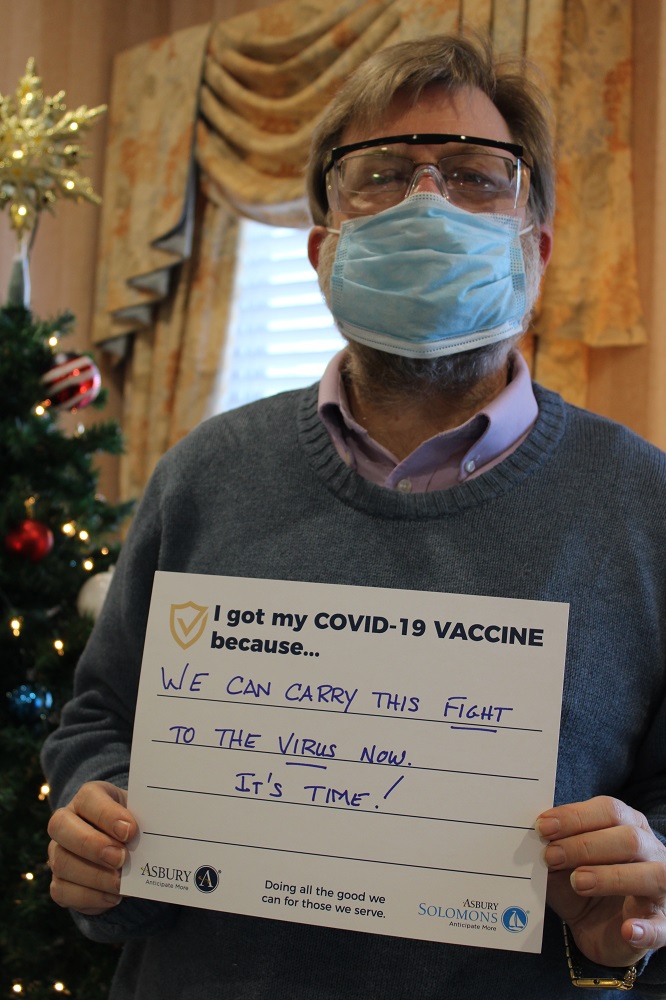
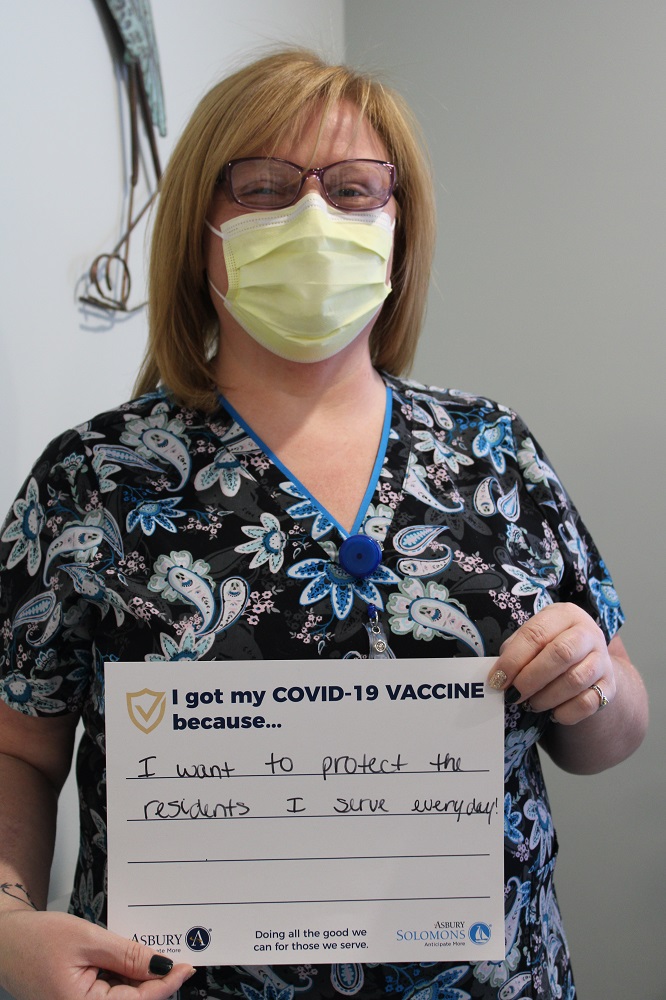
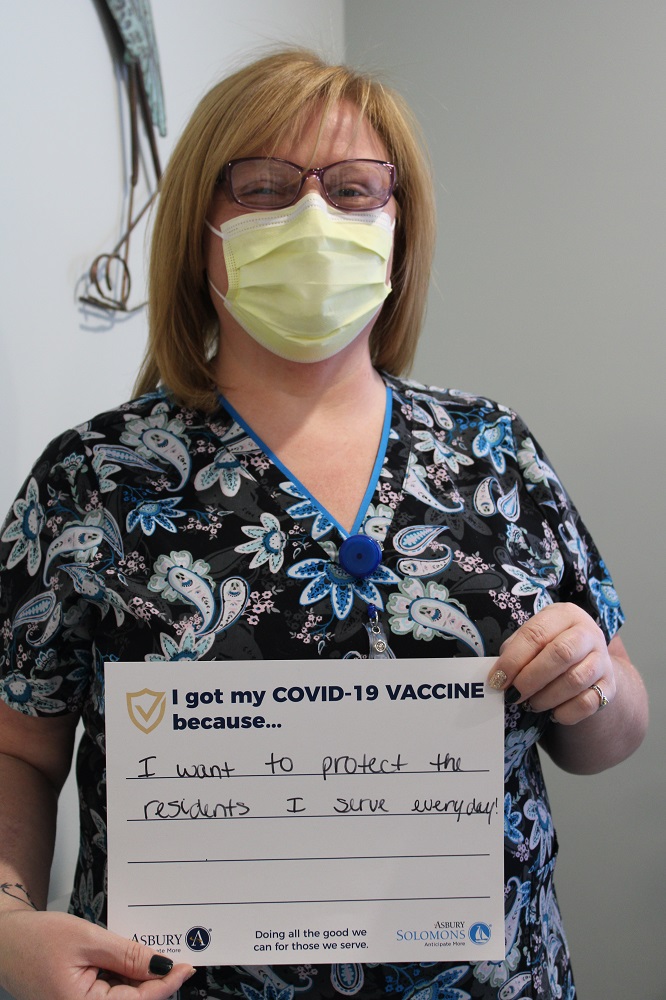


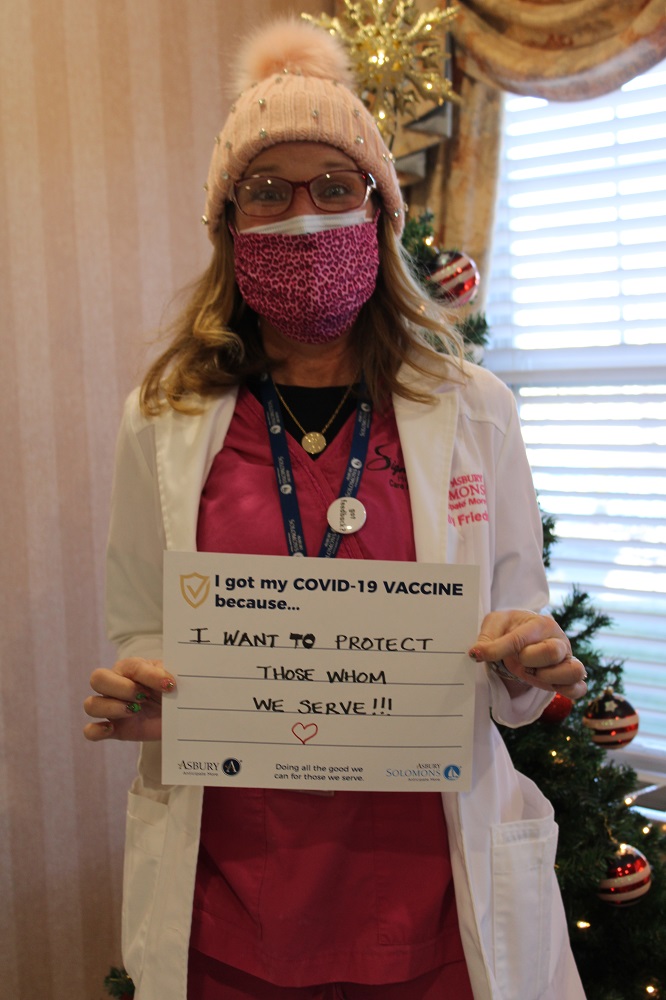

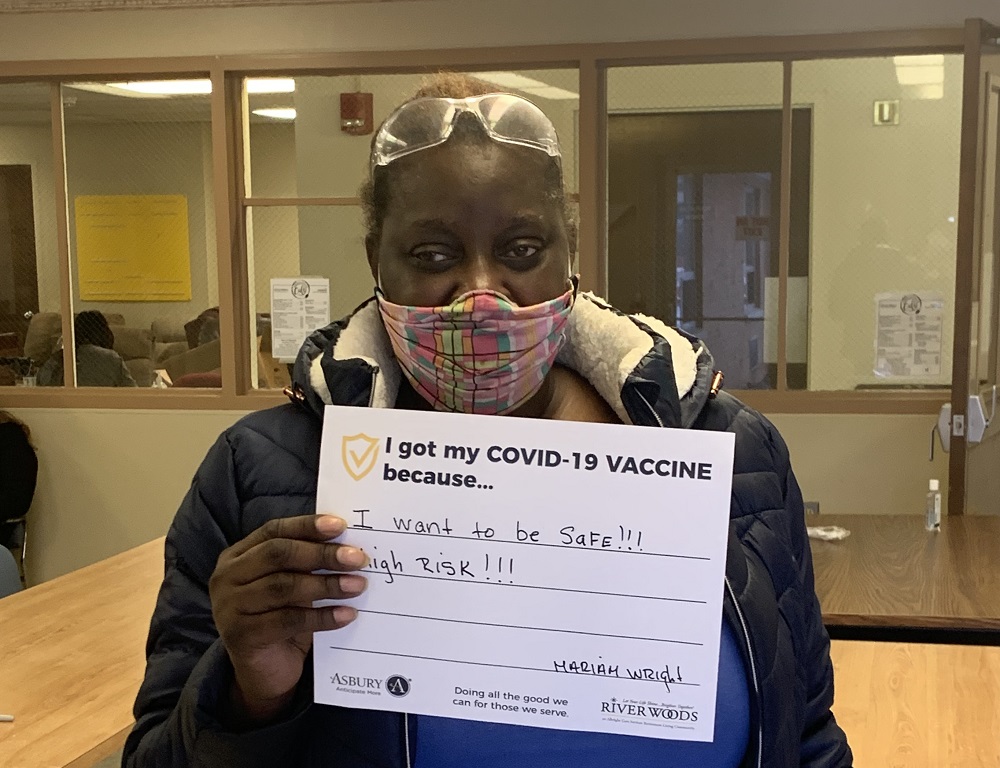









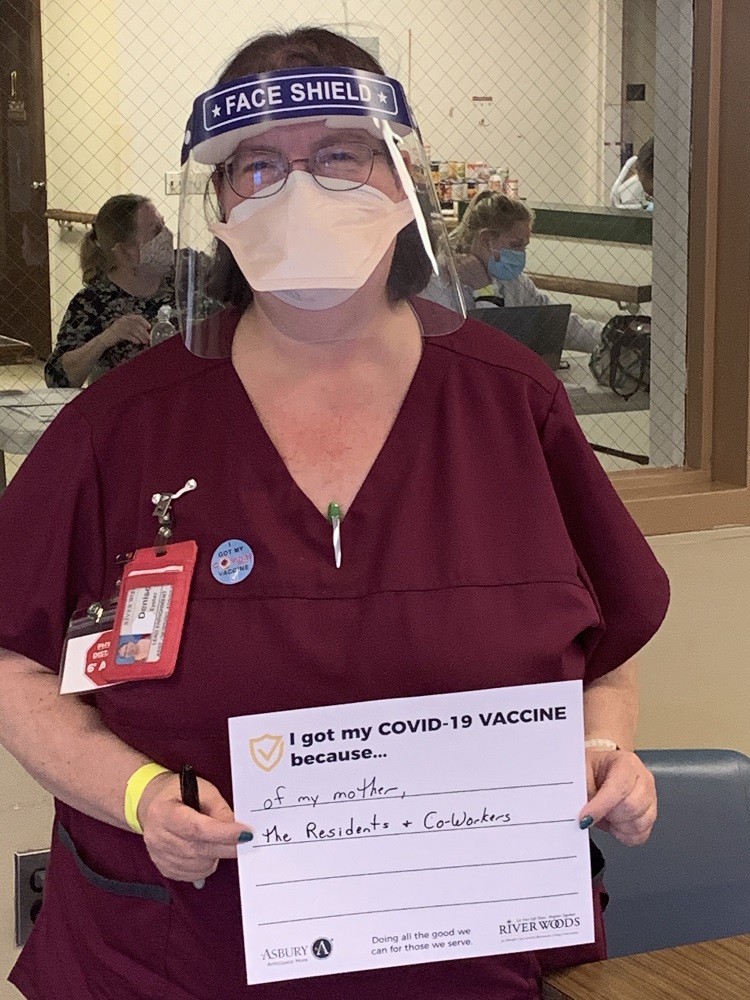
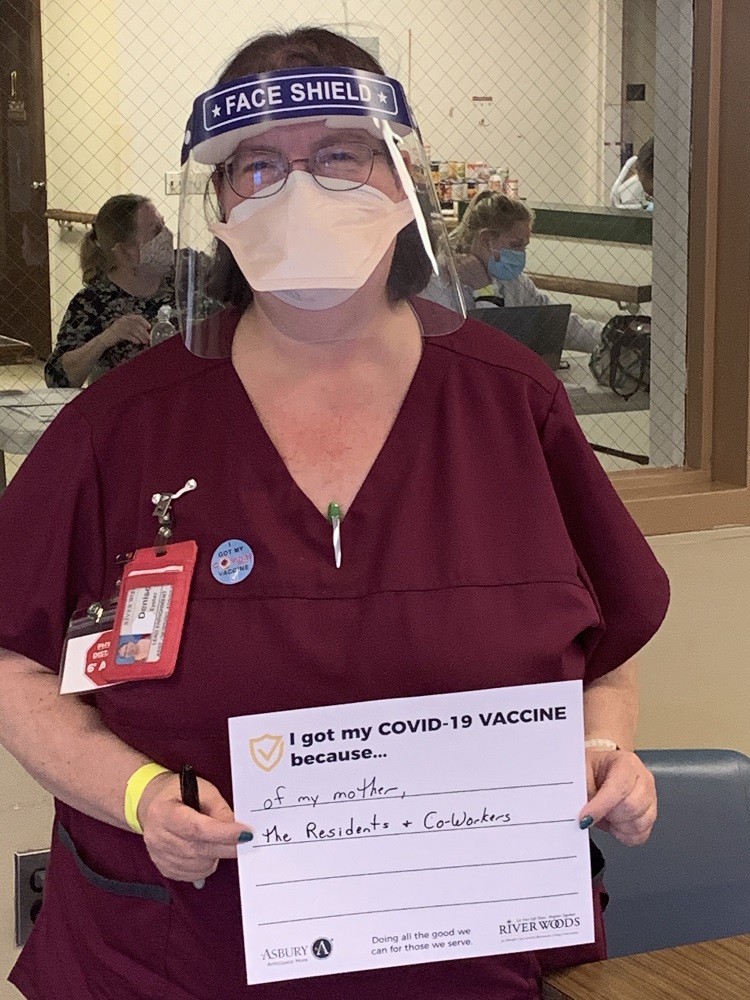




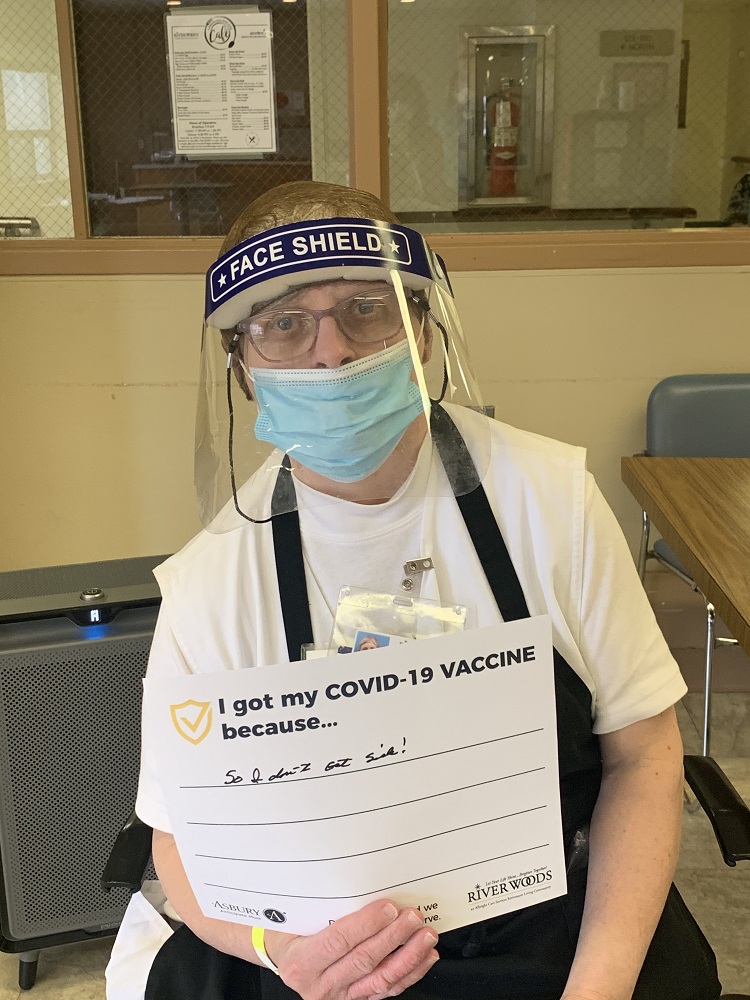













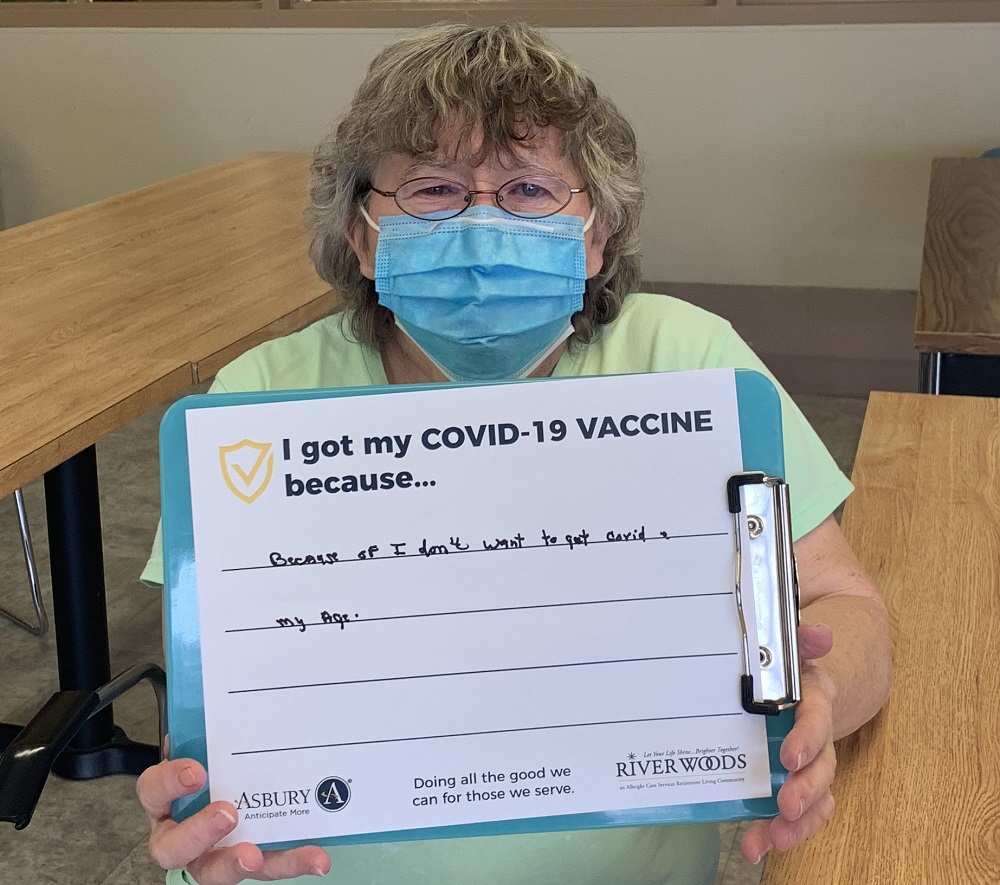



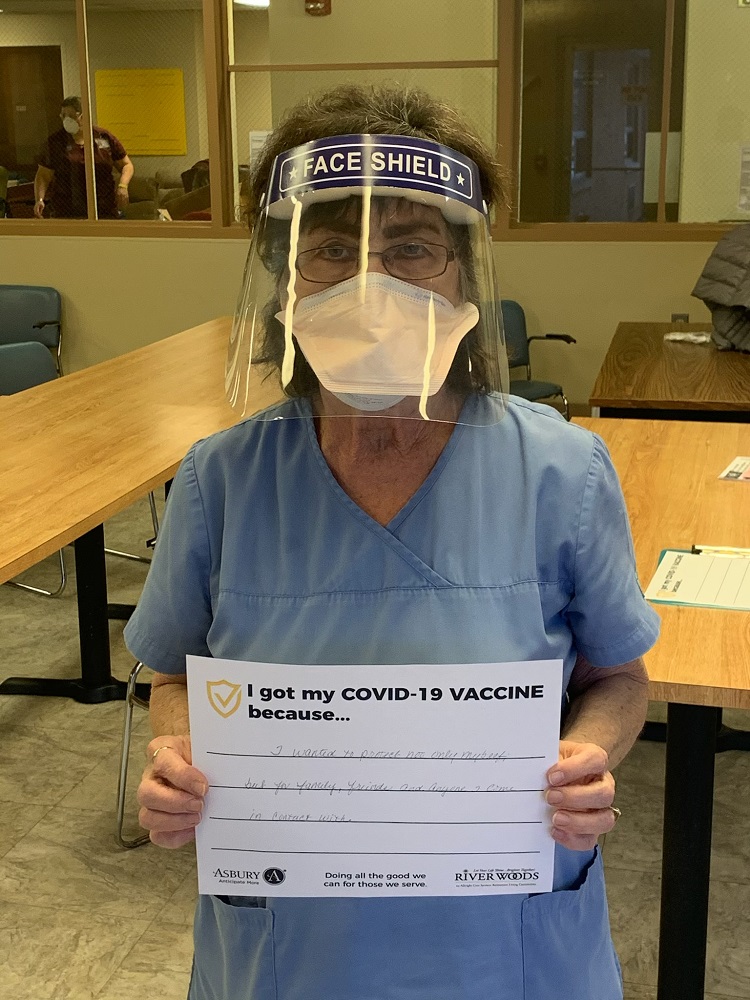













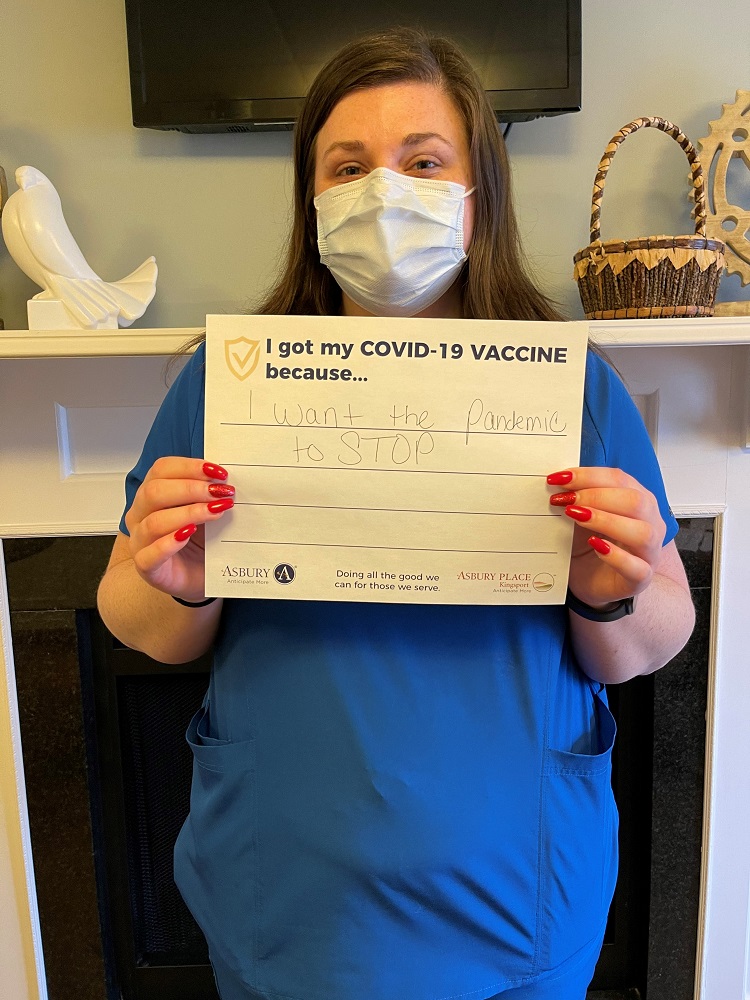



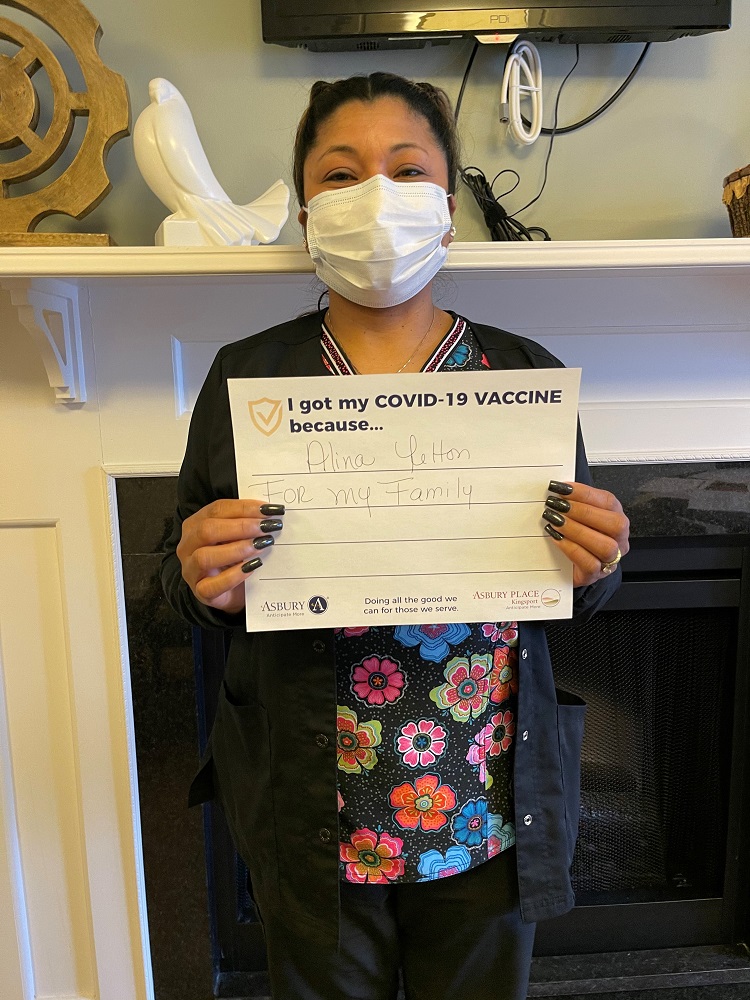
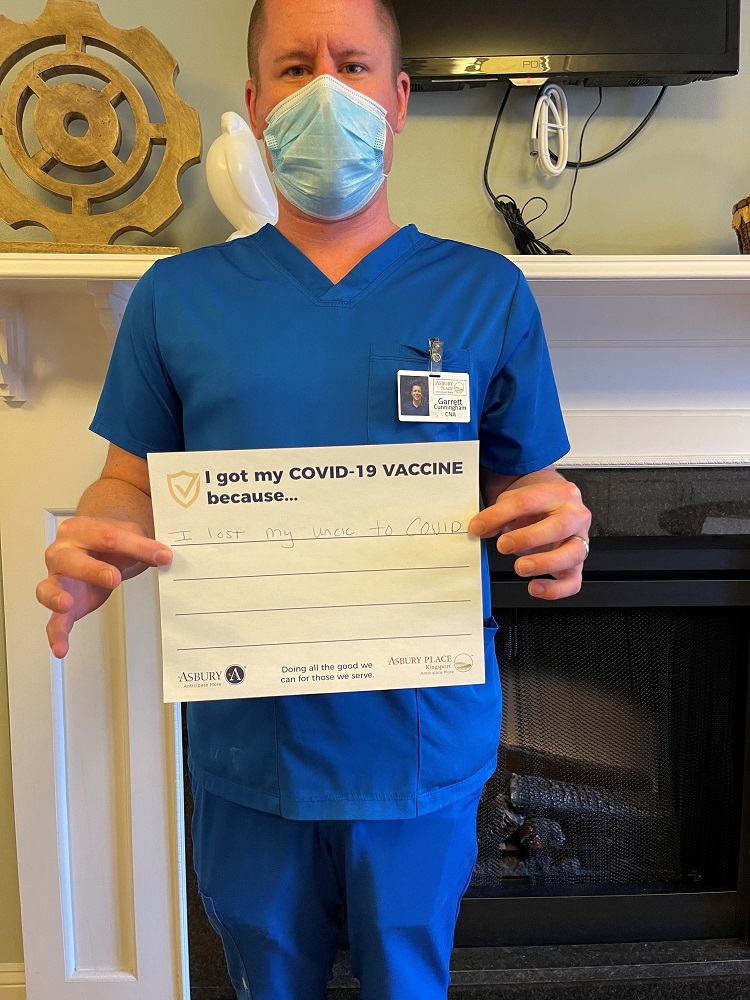











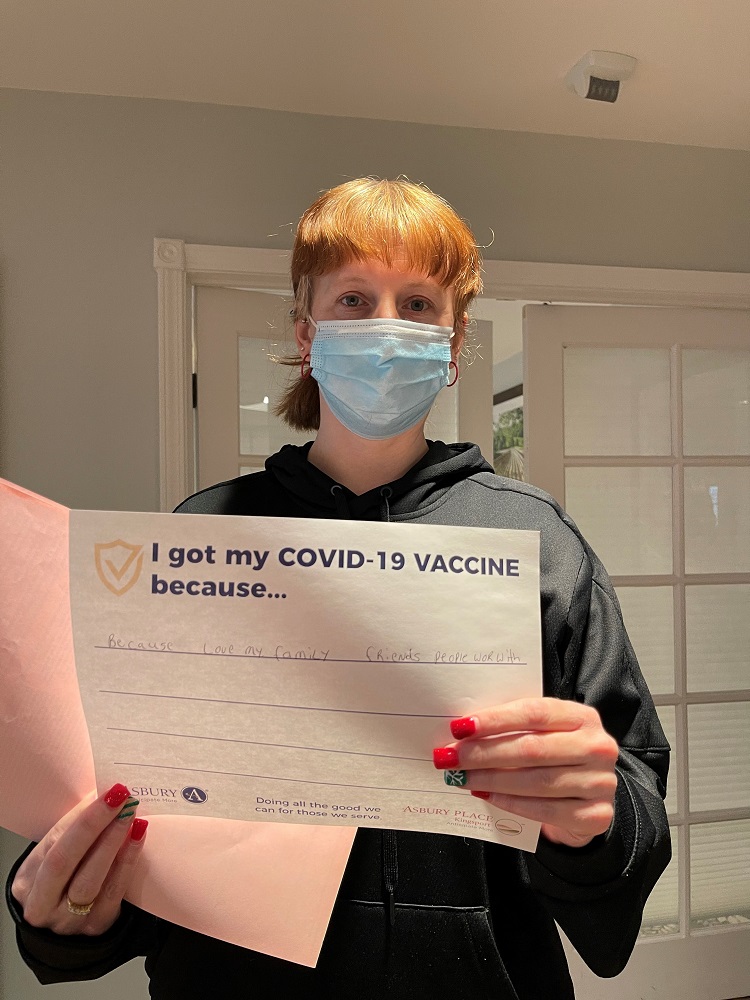





































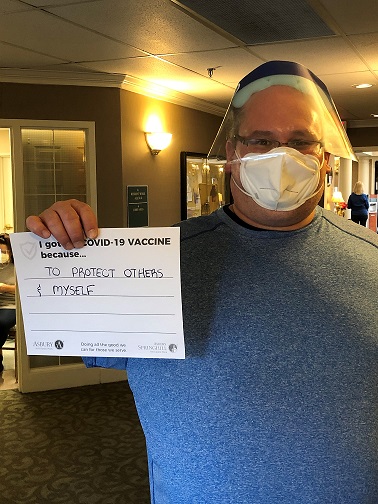










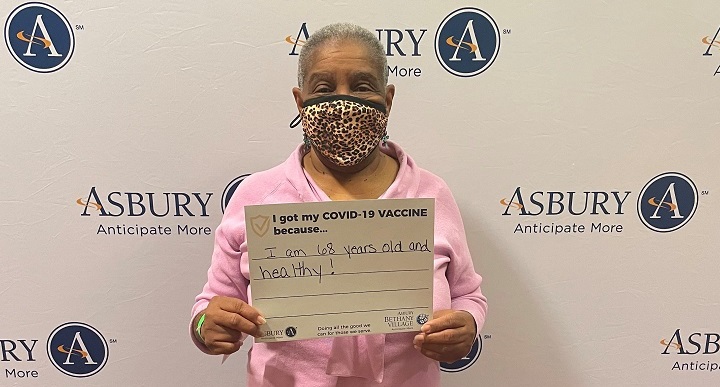
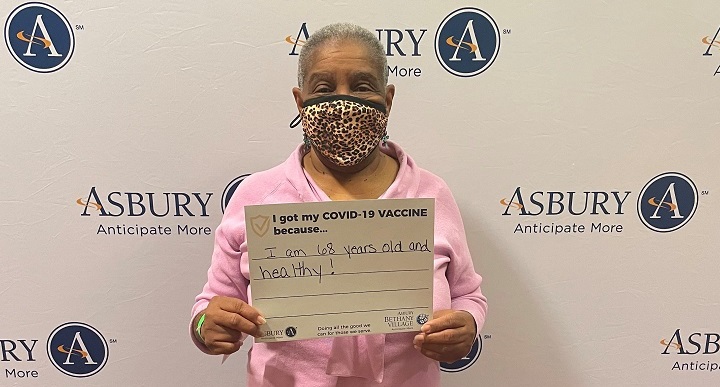
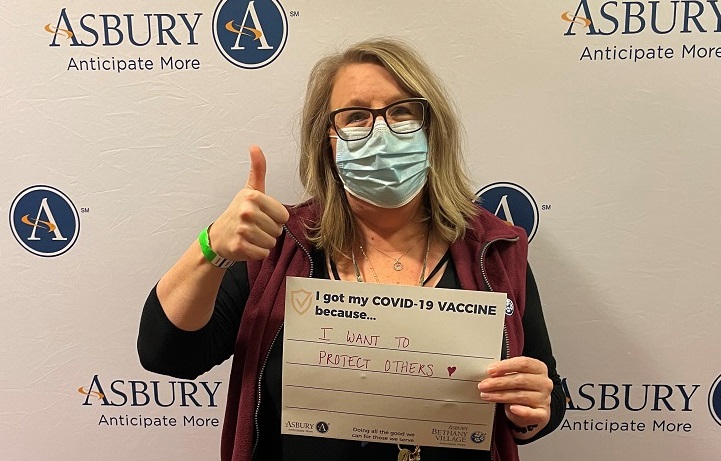




























Vaccine Safety and Other FAQs
*Sources include the CDC, FDA, and Pfizer and Moderna.
What is the vaccination process?
CVS Pharmacy staff will administer the vaccines on site at each Asbury community. Similar to the process for the flu vaccinations, you will need to show your insurance card at the time of vaccination. The vaccine will require two shots administered 21 days apart (Pfizer vaccine) or 28 days apart (Moderna vaccine). You must get the second dose for the vaccine to work.
Currently, the FDA has granted Emergency Use Authorization to the Pfizer and Moderna vaccines.
How was the vaccine tested for safety?
The FDA performs rigorous testing for safety in keeping with the process used for vaccines such as flu. Two independent scientific advisory groups also reviewed data from the COVID-19 vaccine clinical trials. The FDA has concluded that the benefit of receiving the vaccine outweighs the risk of not receiving it for most people.
Vaccine approval requires four clinical trial phases that assess its effectiveness and safety in different populations. A vaccine that has been approved as safe to use has been tested in tens of thousands of people with no significant harmful side effects noted. The current Phase 3 trials have 30,000 to 50,000 participants. The FDA advises a minimum of 3,000 participants to assess safety. Phase 4 involves ongoing monitoring and data gathering over an 8-week period following vaccination, as most adverse side effects have been shown to occur within 6 weeks following vaccination.
Who was included in the Pfizer and Moderna trials?
The Pfizer trials included more than 40,000 people. Of those, 10% were Black and 13% Latino, and 45% were 56 to 85 years old. The Pfizer vaccine is showing a protection rate of 95%.
The Moderna trials included more than 25,000 people. Of those, 10% were Black and 20% were Latino, and 23% were 65 years or older. The Moderna vaccine is showing a protection rate of 94%.
Are there side effects?
The vaccine can cause short-term discomfort such as headache, muscle pains, fatigue, chills, fever and pain at injection site in a percentage of the people who receive them. This is the effect of your body developing immunity – it means the vaccine is working! Most trial participants reported that the discomfort went away after a day, sometimes sooner. The discomfort can be more pronounced when you receive the second shot.
How does the vaccine work and how soon will I be protected?
The vaccine does not contain any active COVID virus and do not cause COVID infection. The vaccine works within our bodies to create an immune response to the presence of COVID-19 should we be exposed to it. It will help prevent the illnesses associated with COVID-19. A person may already be infected with COVID-19 when they get the vaccine but are asymptomatic. If they later have symptoms of COVID-19 or test positive for it, it does not mean they got COVID-19 from the vaccine.
Recipients receive a shot with vaccine material 21 to 28 days apart. You must get the second dose for the vaccine to work. Protection typically begins within 2 weeks after the second shot.
What if I have had COVID-19 already?
Trials have shown that it is safe to get the vaccine if you have already had COVID-19. If you have had a test that shows you have COVID-19 antibodies, the vaccine is still safe for your and may protect you further from future COVID-19 infections.
How long will the vaccine last?
Because the trials are still in the early stages, we will not know this answer for some time. It may be that the COVID vaccine is annual in the same way as the flu.
Resident Information
Asbury will continue to work aggressively for vaccine distribution to residents at all levels of care as well as all associates.
We strongly encourage residents to receive the vaccine. While it is not required, getting the vaccine falls very much in line with our approach throughout the pandemic – that we partner together in doing all that we can to minimize the impact of the coronavirus. Depending on how the evidence of transmission and standards of care evolve, residents who choose not to receive the vaccine may be subject to greater restrictions in the future. Just as we have throughout the pandemic, we are working closely with the leadership teams at each community as we enter this next phase in combating COVID-19. As new information is available, we will share important details with you.
The CDC has developed a phased approach that offers vaccines to Health Care Center/Skilled Nursing and Personal Care/Assisted Living residents and associates as part of Phase 1. To date Phase 1 does not include residents living independently at our communities. Vaccine eligibility, processes, and supplies are set by each state and they vary. Asbury is working aggressively to secure vaccines and to find additional avenues for all those who live and work at our communities and wish to be vaccinated.
Asbury has also called together a team of leaders and caregivers, which includes representation from each community, who will serve as a work group that will steward the entire vaccination program for all residents and associates. Please also reach out to the Executive Director at your community if you have additional questions.
Community Vaccine Schedules
Below are the confirmed vaccination dates with our partner CVS (unless otherwise noted). If changes to confirmed dates are made, updates to the vaccine schedule will be updated and communicated to appropriate parties promptly. Vaccinations require two doses 21 days apart (for Pfizer vaccine) or 28 days apart (Moderna vaccine).
**Note: No one may receive a first vaccine dose on the last clinic date as it requires 2 doses to be effective.
Asbury Methodist Village
- Wilson Health Care Center
- Series A (Pfizer): January 7, January 28, February 18
- Series B (Pfizer): January 8, January 29, February 19
- Kindley Assisted Living:
- Series A (Pfizer): February 9, March 2, March 23
- Residents of Residential Living (65+) at Rosborough Center by Holy Cross Health Partners
- Clinic 1 (Moderna): February 1, February 2, February 3
- Clinic 2 for second dose (Moderna): March 1, March 2, March 3
Asbury Place Kingsport
- Health Care Center
- Series A (Moderna): January 4, February 1, March 1
- Steadman Hill Assisted Living
- Series A (Moderna): January 26, February 23
- Residential Living
- Mac’s LTC Pharmacy Solutions (Moderna): February 4, March 4
Asbury Place Maryville
- Health Care Center
- Series A (Moderna): January 8, February 5, March 5
- Assisted Living
- Series A (Moderna): January 15, February 12, March 12
- Residential Living
- Mac’s LTC Pharmacy Solutions (Moderna): February 3, March 3
Asbury Solomons
- Health Care Center
- Series A (Pfizer): December 26, January 16, February 6
- Assisted Living
- Calvert County Clinic (Moderna): January 9, February 6
Bethany Village
- Skilled Nursing
- Series A (Pfizer): January 14, February 4, February 25
- Assisted Living
- Series A (Pfizer): February 4, February 25, March 18
- Residential Living East Campus
- Care Options Rx Clinic (Moderna): February 7, March 21
- Residential Living West Campus
- Care Options Rx Clinic (Moderna): February 8, March 22
Normandie Ridge
- Health Care Center
- Series A (Pfizer): January 15, February 5, February 26
- Personal Care
- Series A (Pfizer): February 5, February 26
RiverWoods
- Health Care Center
- Series A (Pfizer): January 4, January 25, February 15
- Series B (Pfizer): January 13, February 3, February 24
- Personal Care
- Series A (Pfizer): February 5, February 26
Springhill
- Forest View Health Care Center
- Series A (Pfizer): January 10, January 31, February 21
- Personal Care
- Series A (Pfizer): January 24, February 14, March 7
